Tissue Genomic DNA Kit - Magnetic Beads Based DNA Extraction Kit
This magnetic bead genomic DNA purification kit was designed specifically for isolating the Tissue genomic DNA
Tags: Magnetic Beads / Purification Kits / Extraction Kit / Isolation Kit / Tissue genomic DNA
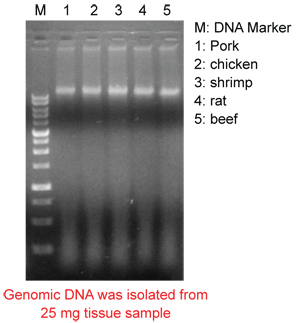
The Tissue Genomic DNA Kit was designed specifically for genomic DNA isolation from tissue samples.
Its special buffer system will efficiently lyse cell and degrade protein, allowing for DNA to be easily bound by the surface of the magnetic beads.
The other non-specific binding particles are removed with a wash buffer, and the genomic DNA is released from the beads by addition of a low ionic strength buffer and heat.
Genomic DNA can be purified manually within 30-50 minutes (using most magnetic separators) or the kit can be easily adapted to satisfy most automated Nucleic Acid purification systems.
• Sample: Up to 30mg of the animal tissue
• Format: Manual or automated genomic DNA isolation
• Operation time: Within 50 minutes (manual)
• Applications: Restriction Enzyme Digestion, Southern Blotting, PCR and qPCR assays
• Storage: Room temperature
Tags: Magnetic Beads / Purification Kits / Extraction Kit / Isolation Kit / Tissue genomic DNA
Final price excl. shipping costs3
- verfügbar / avaílable
- 3 - 5 days for delivery / 3 - 5 Tage Lieferzeit1
Contents
Magnetic Beads: 2 ml
Lysis Buffer: 30 ml
Grind Buffer: 25 ml
Wash Buffer: 80 ml
Release Buffer: 20 ml
Required Materials:
Absolute EtOH
Magnetic separator
1.5 ml microcentrifuge tubes
Proteinase K (10 mg / ml)
Water bath/ Dry bath
Trouble shooting for DNA Extraction with Magnetic beads
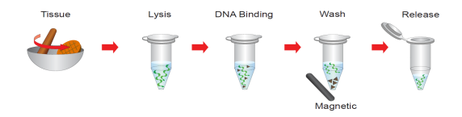
Tissue Genomic DNA Kit Protocol
Sample Preparation
1. Cut off up to 30 mg of the animal tissue and transfer it to a 1.5 ml microcentrifuge tube.
2. Add 200 μl of the Grind Buffer to the tube and continue to homogenize the sample tissue by grinding.
Step 1 Lysis
1. Add 20 μl of the Proteinase K (10 mg/ml) to the sample mixture and mix by vortex.
2. Incubate at 65°C for 30 minutes to lyse the sample. During the incubation, invert the tube every 5 minutes.
3. Centrifuge for 5 minutes at 5,000 x g.
4. Transfer the supernatant to a new 1.5 ml microcentrifuge tube and add 300 μl of the Lysis Buffer.
5. Mix well and incubate at 65°C for 5 minutes. During this time, pre-heat the Release Buffer to 65°C for the Step 4.
6. Add 300 μl of the absolute EtOH to the lysate and mix well.
Step 2 DNA Binding
1. Add 20 μl of the magnetic beads. Mix well by gently shaking for 3 minutes.
2. Place the tube in a magnetic separator for 30 seconds.
3. Remove the solution (If the mixture becomes viscous, increase the magnetic bead separation time).
Step 3 Wash
1. Add 800 μl of the Wash Buffer and mix well (Following the wash, the mixture will no longer be viscous).
2. Place the tube in a magnetic separator for 30 seconds. Remove the solution.
Step 4 Release
1. Add 200 μl of the Release Buffer (pre-heated to 65°C) and mix well.
2. Incubate for 3 minutes at 65°C (During the incubation, shake the tube vigorously every minute).
3. Place the tube in a magnetic separator for 1 minute.
4. Carefully transfer ONLY the clean portion of the solution to a clean tube.
NOTE: Be sure and allow the magnetic beads to disperse completely during the binding, wash and elution steps.
Its special buffer system will efficiently lyse cell and degrade protein, allowing for DNA to be easily bound by the surface of the magnetic beads. The other non-specific binding particles are removed with a wash buffer, and the genomic DNA is released from the beads by addition of a low ionic strength buffer and heat.



