Proteinase K - Lyophilizate and customized solutions
Proteinase K and its solution is used for protease digestion during DNA and RNA preparation. Proteinase K is very active and stable, even at room temperature. Proteinase K is used as component in Diagnostics Kits or in DNA/RNA purification systems.
Proteinase K Lyophilisat product sizes
405-002-25: 4x25 mg
405-002: 200mg
405-005: 500mg
405-010: 1g
405-010L: 5x1 g
405-010XL: 15x1 g
405-010XXL: 30x1 g
Alternative: Proteinase K powder and Storage buffer separately: 1x125 mg + 6,2 ml buffer (50 mM Tris HCl (pH 8.0), 1 mM CaCl2, 50 % Glycerin)
Final price excl. shipping costs3
- verfügbar / avaílable
- 1 - 3 days for delivery / 1 - 3 Tage Lieferzeit1
Description: Proteinase K is a serine protease that exhibits a very broad cleavage specificity. The Protein with a molecular weight 28.900 kD cleaves peptide bonds adjacent to the carboxylic group of aliphatic and aromatic amino acids. Proteinase K is not inactivated by chelating reagents such as EDTA or detergents such as SDS and is active over a wide range of pH (4-12.5).
Activity: > 30 units/mg Lyophilizate and > 40 units/mg protein
Unit definition: Unit definition One unit is the amount of enzyme which releases at 37°C in 1 min as many folin-positive amino acids and peptides from haemoglobin as 1 μmol of tyrosine.
Features:
The protease or - its solution is a highly active and stable protease with low cutting specificity. The enzyme belongs to the group of subtilisine-related serine proteases and is strongly inhibited by PMSF.
Usage: In presence of 0.5 – 1 % SDS Proteinase inactivates DNases and RNases in eukaryotic and microbiological cell cultures. The use of Proteinase K during lysis of the cells allows the isolation of intact highly-molecular nucleic acids.
Quality:
- purified by chromatography and lyophilised
- RNases: not detectable
- DNases: not detectable
- Exonucleases: not detectable
Each batch of Proteinase K is tested for the presence of fungal and bacterial DNA. Moreover, the specific activity assay of each batch of the enzyme is performed.
Storage:
4 °C or -20 °C for at least 24 months, shipment at ambient temperature or blue ice
Proteinase K as Lyophilizate
Datasheet Proteinase K Lyophilisate
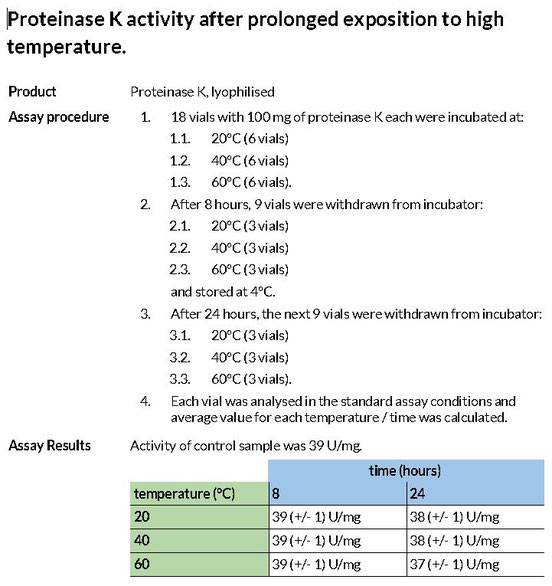
Long term stability test of Proteinase K
Determination of the the stability of Proteinase K during long-term storage at room temperature. The tested Lot was compared to the fresh production batch in 6 month time intervals. Assay conditions described below.
Lot: tested: P040917 control LOT: current production batch
Storage buffer for Proteinase K in solution:
20 mM Tris pH 7,5, 1 mM CaCl2, 0,02% sodium acid, 50% glycerol (v/v).
Test results
Powder / Solution
| Form | control Lot | 6 months | 12 months | 18 months | 24 months |
| Lyophilized form | 100 % | 98 % | 101 % | 98 % | 99 % |
| Solution 20 mM | 100 % | 99 % | 98 % | 98 % | 97 % |
Assay conditions
● Temperature: 37 °C, A750
● Reaction volume: 900 μl
● Substrate Solution: 2% Haemoglobin with 6M Urea pH 7.5
● Spectrophotometer: Eppendorf Biospectrometer
Proteinase K as solution: 20 mg/ml (50 mM Tris HCl (pH 8.0), 1 mM CaCl2, 50 % Glycerin)
Proteinase K powder and Storagebuffer separatly: 1x125 mg + 6,2 ml buffer
Proteinase K solution: as bulk or in customized packsizes and concentration
Final price excl. shipping costs3
- verfügbar / avaílable
- 1 - 3 days for delivery / 1 - 3 Tage Lieferzeit1
Proteinase K as solution
Datasheet Proteinase K Solution 20 mg/ml
Custom aliquoting service
GeneON provides customized amounts of Proteinase K Powder and storage buffer on request. We precisely dispense Prot.-K aliquots (liquid/lyophilized) that will fit the format of your choice.
Free sample request for Proteinase
Dear customer, GeneON likes to send free samples of to convince the valued customer about the quality. Please understand that the shipping costs may be very high to some destinations. That is the reason why we cannot assure to fulfil all sample requests. Sorry for that, please understand. You may also set an inquiry/order directly by e-mail .
Material Safety Datasheet
Material Safety Datasheet
Deutsche Beschreibung: Proteinase K
Related products
Proteinase K
|
References: https://www.researchgate.net/publication/365110345_Investigation_of_the_Thermal_Stability_of_Proteinase_K_for_the_Melt_Processing_of_Poly_l_-lactide
|