T 4 DNA Ligase (Weiss-units)
T 4 DNA Ligase high concentrated in Weiss units. T4 DNA Ligase catalyzes the formation of a phosphodiester bond between juxtaposed 5´ phosphate and 3´ hydroxyl termini in duplex DNA or RNA (Delivered in Weiss Units)
Applications:
- Cloning of restriction fragments
- joining linkers and adapters to blunt-ended DNA
- gene (gene fragments) synthesis.
Description:
T4 DNA Ligase catalyzes the formation of a phosphodiester bonds between 5' phosphate and 3' hydroxyl termini in duplex DNA/RNA. This enzyme can join blunt end and cohesive end termini, repair
single stranded nicks in duplex DNA, RNA, or DNA/RNA hybrids.
Concentration: 2,5 Weiss Units / µl ( 500 CE units / µl)
Source:
Isolated from E.coli strain that carries the cloned DNA ligase gene from bacteriophage T4
Usage:
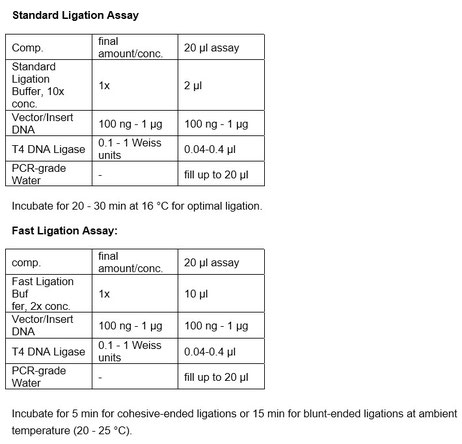
For most cohesive end ligations, a 30 minute incubation at 20°C is sufficient. Incubations at 16°C for 4-16 hours are routinely used for the majority of applications.
Ligation of blunt ends and single-base pair overhang fragments requires more enzyme to achieve the same extent of ligation as cohesive end DNA fragments. Ligation may be enhanced by addition of
PEG, or by reducing the ATP concentration. ATP is an essential cofactor for the reaction.
Storage buffer:
10 mM Tris-HCl pH 7.4, 50 mM KCl, 0.1 mM EDTA, 1 mM DTT, 200 μg/ml BSA and 50 % [v/v] glycerol)
Unit definition:
One unit is defined as the amount of enzyme required to give 50% ligation of Hind III fragments of lambda DNA in 30 minutes at 16°C at 5' termini concentration of 0.12 µM (300 µg/ml). One
Weiss unit equals 200 Cohesive End Ligation Units.
Reaction buffer (10X):
500 mM Tris-HCl pH 7.8 at 25 °C, 100 mM MgCl2, 100 mM DTT, 10 mM ATP and 25 μg/ml BSA.
FAST LIGATION BUFFER (2x): 60 mM Tris-HCl pH 7.8 at 25 °C, 20 mM MgCl2, 20 mM DTT, 2 mM ATP and 10 % PEG
Reaction buffer "Fast Ligation" (2X): 60 mM Tris-HCl pH 7.8 at 25 °C, 20 mM MgCl2, 20 mM DTT, 2 mM ATP and 10 % PEG
Quality Assurance:
Free of contaminating exonuclease and endonuclease
Storage: shipped on blue ice, 24 months at -20°C
Notes for T4 DNA Ligase
- One Cohesive-End Ligation Unit (CEU) is defined as the amount of enzyme required to give 50 % ligation of Hind III fragments of λ DNA (5' DNA termini concentration of 0.12 μM, 300 μg/ml) in a total reaction volume of 20 μl in 30 minutes at 16 °C in 1x T4 DNA Ligase Reaction Buffer.
- One Weiss unit is equivalent to approx.200 CEU.
- T4 DNA Ligase is strongly inhibited by NaCl or KCl if the concentration exceeds 200 mM.
- Ligation of blunt-ended and single-base pair overhang fragments requires about 50 times as much enzyme to achieve the same extent of ligation as cohesive-end DNA fragments. Blunt-end ligation may be enhanced by addition of PEG 4000 (10 % w/v final concentration) or hexamine chloride, or by reducing the ATP concentration to 50 μM.
- To dilute T4 DNA Ligase for subsequent storage at -20 °C a storage buffer containing 50 % glycerol should be used; to dilute Ligase for immediate use, 1x Reaction Buffer is recommended.
T 4 DNA Ligase high concentrated catalyzes the formation of a phosphodiester bond between juxtaposed 5´ phosphate and 3´ hydroxyl termini in duplex DNA or RNA
* availibility of sample size may be limited
Final price excl. shipping costs3
- verfügbar / avaílable
- 1 - 3 days for delivery / 1 - 3 Tage Lieferzeit1
Datasheet T4 DNA Ligase
Material Safety Datasheet

References / Protocols / Notes / Recomendations / Tests